The Galvano-Fenton Advanced Oxidation Process
The Fenton Reaction
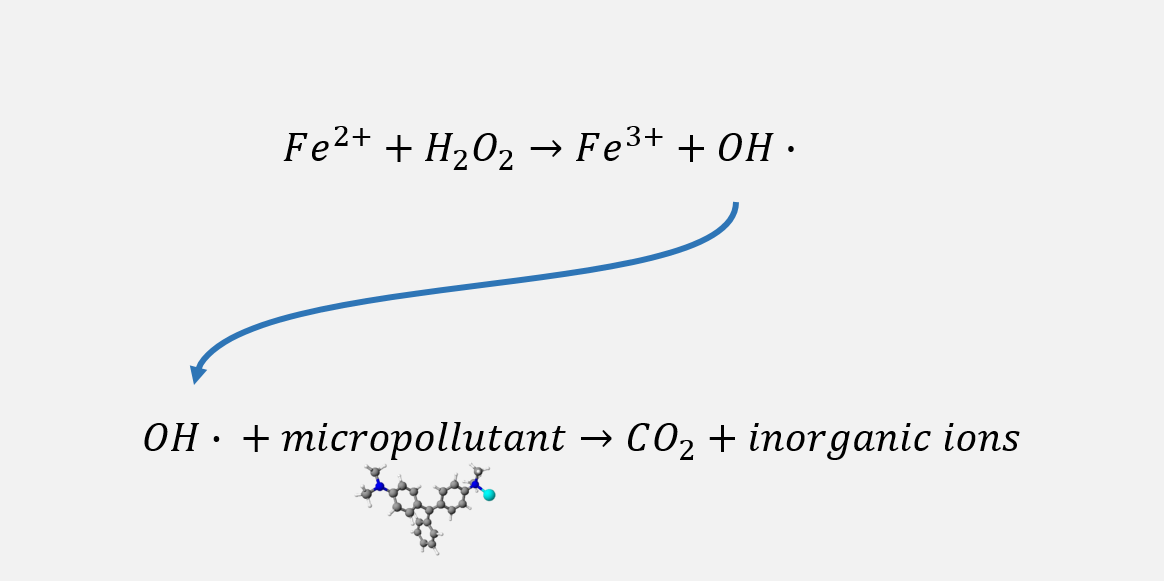
Fenton reaction describes the formation of hydroxil radical OH• , a very strong oxidant chemical compound, from Hydrogen Peroxide H202. This reaction is catalyzed by metallic ions, the most famous one being Iron (II), that gets oxidized with the following reaction :
Fe2+ + H202 → OH• + OH- + Fe3+
This reaction is very effective at acidic pH (around 2-3). The presence of a very unstable strong oxidant like OH• can be used to mineralize molecular organic pollutants (such as pecticides, drug residues, etc.), whom impact on water quality constitutes one major present and future challenge for environmental and public health.
Coupling Fenton reaction with galvanic corrosion
One of the main challenges in using the Fenton technology is the constant need for reagent supply (Iron and H2O2). Galvanic corrosion allows to supply Fe ions from a sacrificial anode, at constant rate and without the use of electricity, by connecting the Fe anode with a more noble metallic cathode (Cu for example). With this system, Only H2O2 has to be supplied,
More interestingly, copper cathode can be replaced with air carbon electrode, which selectively reduce O2 into H2O2 with the following semi-reaction :
O2 + 2H+ + 2e- → H2O2
With that technology, all reageants are provided without the use of energy. Iron waste could also be used as sacrificial anodes, in order to improve the Life Cycle Assessment
Project
The IronTech ANR project has been developed since Feb. 2023 to evaluate the feasibility of Galvano-Fenton reaction in sea water to treat algae
Related publications
No related publications yet.